JENS MUHLE
Research Interests
- AGAGE (Advanced Global Atmospheric Gases Experiment) (data portal)
AGAGE Steering Committee (2024) - Stratospheric Ozone Depletion, Ozone Depleting Substances (ODSs)
- Global and regional emissions, loss processes, and lifetimes of ODSs and other trace gases
- Top-down (measurement based) verification of bottom-up emission estimates of ODSs and other trace gases
- Top-down (measurement based) verification of compliance with the Montreal Protocol on Substances that Deplete the Ozone Layer and its Amendments
- Halogenated trace gases
- Perfluorinated Compounds (PFCs) and their emissions from global aluminium, rare-earth, and semi-conductor industries
- Trace gas measurement techniques (GC-FID/ECD, GC/MSD, GC/TOF-MS, PTR-TOF-MS, CRDS)
- Atmospheric chemistry
- Wildfire emissions
- Long-range transport of pollutants
Degrees
- Diploma in Chemistry, University of Wuppertal
- Doctor of Natural Sciences, Max Planck Institute for Chemistry, Mainz and Johannes Gutenberg University Mainz, Germany
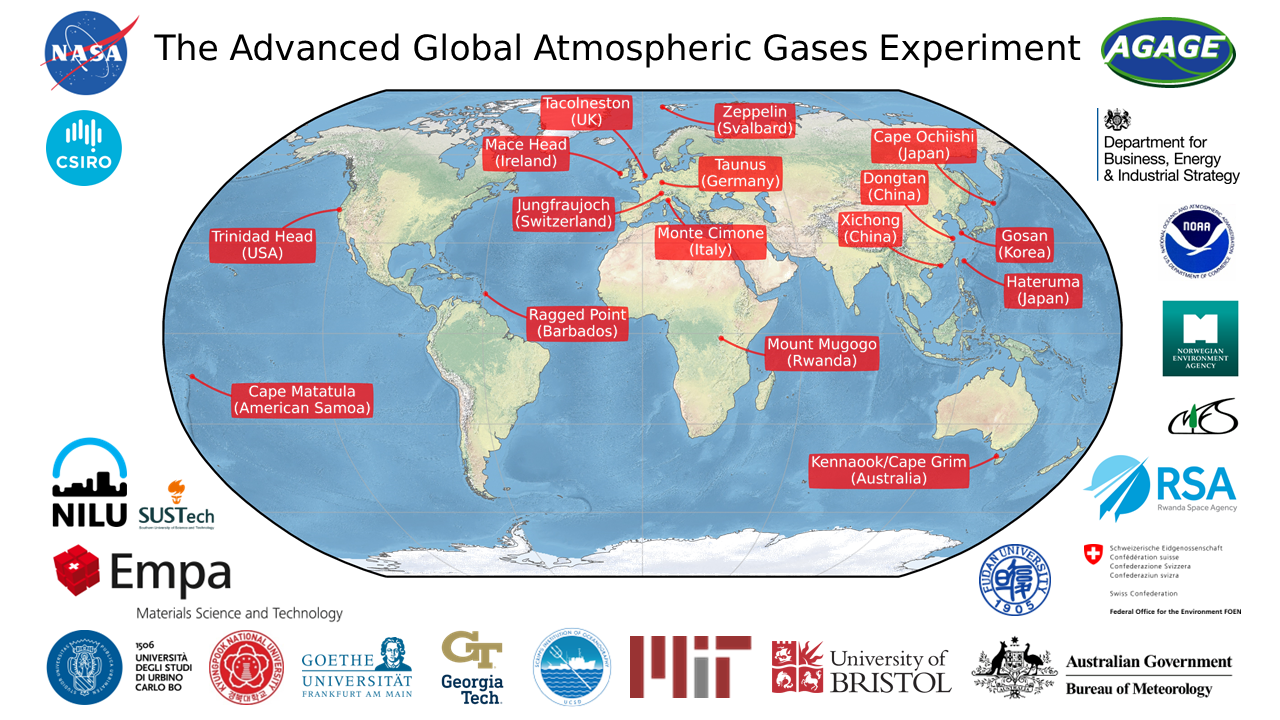
The Advanced Atmospheric Gases Experiment (AGAGE)
AGAGE has been monitoring the global atmosphere since the late 1970s
AGAGE measures most synthetic and natural ozone depleting substances (ODSs) and many other trace gases at 13+ sites around the world. Measurement sites are operated by SIO and many international partners. The central AGAGE experimental and calibration laboratory is located at Scripps Institution of Oceanography at the University of California, San Diego.
Stratospheric Ozone Depletion and the Montreal Protocol
After scientists discovered that the stratospheric ozone layer was being depleted over the Antarctic each austral spring at alarming rates, aka the “ozone hole”, the international community acted and signed the Montreal Protocol on Substances that Deplete the Ozone Layer in the late 1980s. The Montreal Protocol is an international treaty that protects the ozone layer by reducing production and consumption (use) of ozone depleting substances (ODSs), which leads to reduced ODS emissions into the atmosphere. The Montreal Protocol has been strengthened several times over the decades and has been ratified by more than 190 countries.
In very simple terms, stratospheric ozone depletion starts when trace gases that contain chlorine and bromine atoms, such as the Chlorofluorocarbons (CFCs) and Halons, reach the stratosphere and interact at very low temperatures with particles and ultraviolet light during austral spring. Then catalytic cycles involving chlorine and bromine atoms destroy ozone efficiently, allowing more ultraviolet radiation to penetrate deeper into the atmosphere, threatening the health of humans, animals and crops (Scientific Assessment of Ozone Depletion: 2022 and Twenty Questions and Answers About the Ozone Layer: 2022 Update).
By reducing production and use of ODSs the Montreal Protocol has dramatically limited stratospheric ozone depletion and the Montreal Protocol is considered one of the most successful international environmental treaty.
AGAGE has monitored the global atmosphere for ODSs and other trace gases since the late 1970s, providing insights into the amounts of ODSs emitted over time and where they are emitted. This helps to track the progress made in protection the ozone layer and the implementation of the Montreal Protocol.Chlorine and bromine from ozone depleting substances (ODSs)
To understand ozone depletion in the stratosphere, it is important to understand the amounts of chlorinated and brominated compounds emitted into the lower atmosphere (troposphere) as they supply the chlorine and bromine which eventually cause catalytic ozone depletion in the stratosphere (Twenty Questions and Answers About the Ozone Layer: 2022 Update).
AGAGE measurements of ODSs in the global atmosphere provide insights into the amounts of chlorinated and brominated species that can reach the stratosphere and cause ozone depletion.
Equivalent chlorine (ECl) is a measure of how much chlorine and bromine is contained in halogenated compounds which are present in the troposphere. Equivalent chlorine is important to understand how much of these ozone depleting substances (ODSs) can reach the stratosphere and contribute to ozone depletion. Many ODSs are man-made and are emitted into the troposphere due to industrial activities. ECl is based on the abundances of all halogenated ODSs measured by AGAGE.
The use of methyl bromide (CH3Br) as fumigant (against pests) has been substantially reduced in recent years so that most of its present impact is now due to natural sources. Methyl chloride (CH3Cl) is largely emitted from natural sources, thought some anthropogenic sources are known.
(Note, that the impact of brominated and chlorinated very short-lived substances (VSLS), such as dibromomethane (CH2Br2), cannot be adequately reflected in the ECl plot, but they significantly contribute to ozone depletion due to emissions in regions where fast convective processes occur, which means they can reach the stratosphere despite their overall short atmospheric lifetime. AGAGE measures several of these VSLSs and some of the measurement records are shown below.)
Equivalent effective stratospheric chlorine (EESC) is a measure of how much chlorine and bromine (originating from the ozone depleting substances (ODSs) in the troposphere) is available for ozone depletion in the stratosphere.
Due to the phase-out of production and consumption of (most) Ozone Depleting Substances (ODSs) mandated by the Montreal Protocol on Substances that Deplete the Ozone Layer, EESC is slowly decreasing. This slow decrease is projected to eventually allow the ozone layer to recover and prevent the annual Antarctic (and the occasional Arctic) "ozone hole" from forming.
The process is slow and needs to be monitored to detect new emissions of ODSs, which could threaten this recovery. Examples of threats to ozone layer recovery are the recently discovered new CFC-11 emissions in East Asia, continued emissions of carbon tetrachloride (CCl4) and the increasing emissions of several VSLS (stemming from their use as solvents and/or feedstock to produce other chemicals). Note that some ODS have very long atomospheric lifetimes, e.g. the CFCs, which explains their continued large contribution to EESC despite relatively small present emissions.
Radiative forcing
Since many ozone depleting substances (ODSs) are also strong greenhouse gases, the phasing out of ODSs mandated by the Montreal Protocol also slowed down climate change. The impact of greenhouse gases in the atmosphere on climate is often expressed using its radiative forcing (RF). RF basically expresses how much more (or less) energy (from the sun) is trapped in the atmosphere due to the presence of a certain greenhouse gas compared to the pre-industrial atmosphere. As shown below, total radiative forcing (RF, W/m2), or the impact on the climate, of all greenhouse gases (GHGs) is dominated by carbon dioxide (CO2), followed by methane (CH4), the sum of all ozone depleting substances (ODSs), nitrous oxide (N2O), and the sum of all synthetic GHGs.
While most ODSs contain chlorine and/or bromine atoms, it should be noted that nitrous oxide is also a strong ODS. ODSs are causing stratospheric ozone depletion including the "ozone hole" in the Southern Hemisphere every austral spring (see Basic Ozone Layer Science). Once the negative impact of man-made ODSs on the ozone layer was uncovered, the world came together to limit their production and consumption via the Montreal Protocol on Substances that Deplete the Ozone Layer. Several Amendments made the Montreal Protocol stronger, phasing out more compounds (CFCs, halons, certain chlorinated solvents, methyl bromide, ...), more agressively, always allowing developing countries more time than developed countries. Hydrochlorofluorocarbons, which initially replaced CFCs in many application, were eventually also added to the Montreal Protocol. HCFCs have now been phased out in developed countries and they are currently being phased-out in developing countries.
Several ozone depleting substances (ODSs) are also strong GHGs, foremost the chlorofluorocarbons (CFCs). The HCFCs are less harmful to the climate, but they still affect the ozone layer, which is why they are also being phased-out. While HFCs are generally less strong GHGs than CFCs, their contribution to RF is increasing rapidly due to their widespread use and emissions. As shown below, while the impact of the HCFCs on RF has plateued recently (thanks to the Montreal Protocol), the impact of the HFCs on RF continues to grow.
In summary, once CFCs were found to be destroying the stratospheric ozone layer, they (and other compounds) have been phased out by the Montreal Protocol. In many applications CFCs were replaced by HCFCs, later by HFCs, sometimes by other compounds or other means. This replacement protected the ozone layer and also had a positive impact of the climate. Initially a reduction in combined radiative forcing (RF) was achieved, but the sharp increase of the HFCs in the atmosphere threatens to erode this benefit to the climate. This prompted the addition of HFCs to the Montreal Protocol in the Kigali Amendment and their phase-out has now begun.
Total radiative forcing (RF) (left top) and contribution from individual compounds classes: CO2 (carbon dioxide) (left middle), CH4 (methane), (Total) ODSs (ozone depleting substances), N2O (nitrous oxide), and (Total) Synthetic GHGs (left) (left bottom).
(Total) ODS as the sum of RF from CFCs (chlorofluorocarbons), HCFCs (hydrochlorofluorocarbons), solvents (chlorinated solvents and feedstock compounds), and halons (brominated fire fighting compounds) (right top).
Total Synthetic GHGs as the sum of RF from PFCs (perfluorocarbons), HFCs (hydrofluorocarbons), SF6 (sulfur hexafluoride), SO2F2 (sulfuryl fluoride), NF3 (nitrogen trifluoride) (right bottom).
This plot and the following stack plots were produced by CSIRO, one of our many international partners.
Contribution of individual compound groups to total radiative forcing (RF) on a logarithmic scale: CO2 (carbon dioxide), CH4 (methane), (Total) ODSs (ozone depleting substances), CFCs (chlorofluorocarbons), N2O (nitrous oxide), HCFCs (hydrochlorofluorocarbons), CH3CCl3, CCl4 , Kyoto Protocol Synthetics, PFCs (perfluorocarbons), HFCs (hydrofluorocarbons), SF6 (sulfur hexafluoride), halons (brominated fire fighting compounds), SO2F2 (sulfuryl fluoride), NF3 (nitrogen trifluoride).
Radiative forcing (RF) from CFCs (chlorofluorocarbons), HCFCs (hydrochlorofluorocarbons), HFCs (hydrofluorocarbons), and other synthetic greenhouse gases (SGHGs, SF6 (sulfur hexafluoride), PFCs (perfluorocarbons), halons, CH3CCl3 and CCl4).
Several CFCs still contribute significantly to total RF, even though their production and consumption has been phased out by the Montreal Protocol on Substances that Deplete the Ozone Layer. This lasting contribution of CFCs to RF is in part due their long atmospheric lifetimes and in part due to large "banks" (old insulation foams, refrigerators, etc.), from which these compounds are still leaking into the atmosphere. The growth in the contribution of HCFCs to RF has slowed down as consumption and production of HCFCs are being phased out by the Montreal Protocol on Substances that Deplete the Ozone Layer (which mandates a 100% HCFC reduction in developed countries by 2020 and a 67.5% HCFC reduction in developing countries by 2025 and 100% by 2030). In contrast, the contribution of HFCs to RF is increasing unabated, which is why HFCs have recently been added to the Montreal Protocol on Substances that Deplete the Ozone Layer in the Kigali Amendment.
AGAGE global measurements of CFCs
Measurement data for these and other ODSs and GHGs can be found here (data portal).
CFCs have contributed to stratospheric ozone depletion and their use is the main reason for the annual Antarctic "ozone hole". Therefore, production and consumption of CFCs have been phased-out by the Montreal Protocol on Substances that Deplete the Ozone Layer to reduce the size of annual Antarctic "ozone hole" and to allow the ozone layer to eventually recover. Large "banks" of CFCs still exist, for example in old refrigerators and insulation foams where CFCs were used. Moreover, several CFCs have very long atmospheric lifetimes, so that they are only very slowly removed from the atmosphere.
AGAGE global measurements of chlorinated species
Measurement data for these and other ODSs and GHGs can be found here (data portal).
Production and consumption of CH3CCl3 and CCl4 have been phased-out by the Montreal Protocol on Substances that Deplete the Ozone Layer to reduce the size of annual Antarctic "ozone hole" and to allow the ozone layer to eventually recover. CH3CCl3 has been a real success, it has almost disappeared from the atmosphere due to its short atmospheric lifetime and reduced emissions, but many sources of CCl4 emissions still exist as evident from a slower than expected atmospheric decline.
The other compounds shown below are not regulated by the Montreal Protocol on Substances that Deplete the Ozone Layer due to their short atmospheric lifetime and/or predominantly natural original. However, atmospheric abundances of CHCl3 and CH2Cl2,
two chlorinated very short-lived substances (VSLS), are on the rise, most likely due to industrial activities (solvents, feedstocks, etc.).
Note, PCE data is reported on a calibration scale adopted from the National Oceanographic and Atmospheric Administration ( NOAA HATS).